Research Progress of Inorganic Nano-Catalyst for Chemodynamic Therapy
-
摘要: 化学动力学疗法(CDT)是一种侵袭性小的新兴肿瘤治疗方式. 其通过将纳米催化剂引入肿瘤治疗中,促进肿瘤内产生特定毒性的活性氧,从而杀死肿瘤细胞. 从化学动力疗法的原理,无机纳米催化材料的选择,CDT协同光热治疗、化疗、光动力学治疗、放疗共同提升肿瘤治疗效率这几个方面进行综述,说明CDT有望成为一种新型的替代性的肿瘤治疗方式.
-
关键词:
- 化学动力学疗法(CDT) /
- 纳米催化剂 /
- 协同治疗 /
- 芬顿反应
Abstract: Chemodynamic therapy (CDT) is a minimally aggressive emerging cancer treatment, which introduces nanocatalysts into cancer treatment to promote the production of specific toxic reactive oxygen species (ROS) in tumors, inducing the death of tumor cells. Summaried from the aspects of the mechanism of CDT, the choice of nanocatalytic materials, and combinations of CDT with photothermal therapy, chemotherapy, photodynamic therapy and radiotherapy to improve the efficiency of tumor treatment, it indicates that the CDT is expected to become an alternative method of tumor treatment.-
Key words:
- chemodynamic therapy(CDT) /
- nanocatalysts /
- synergistic treatment /
- Fenton reaction
-
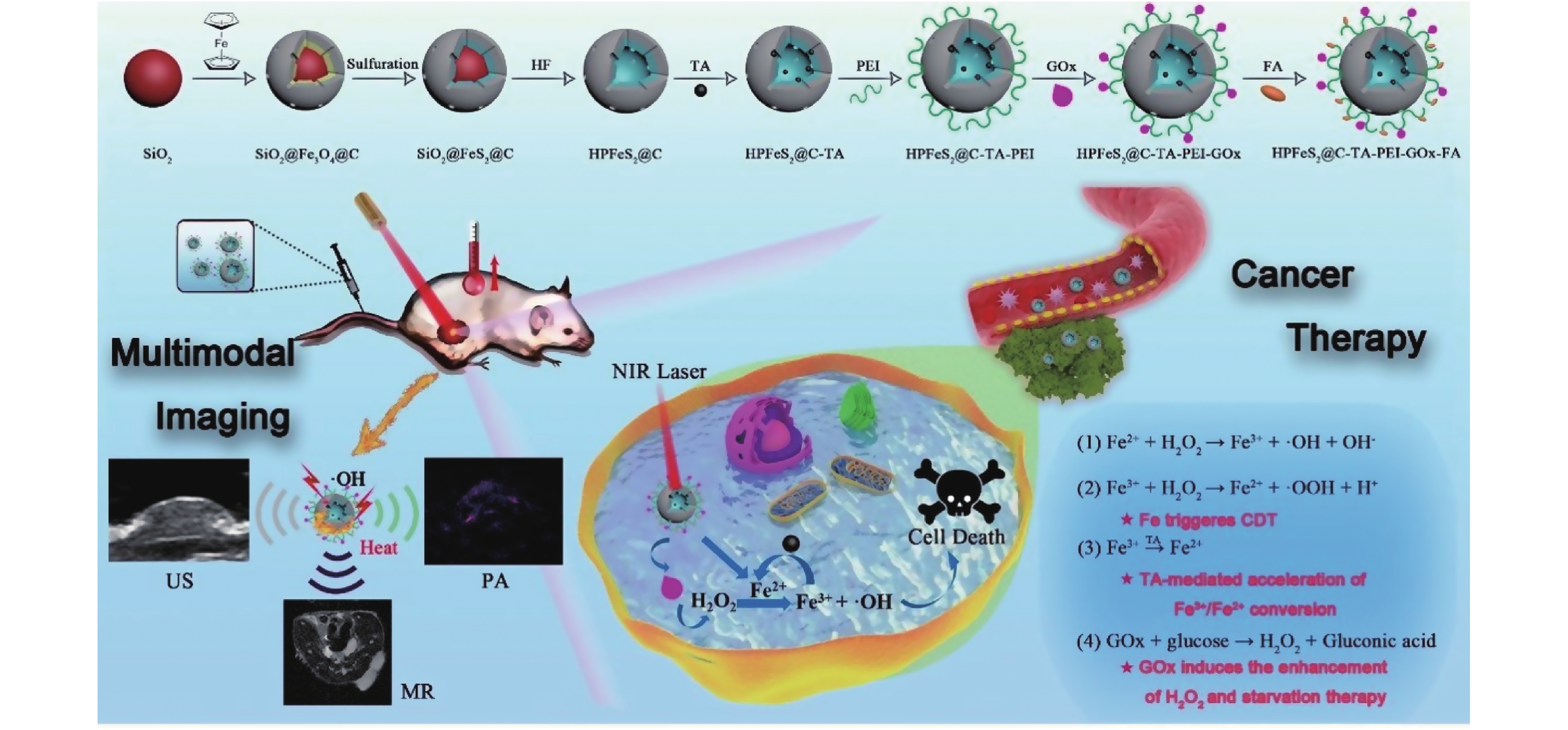
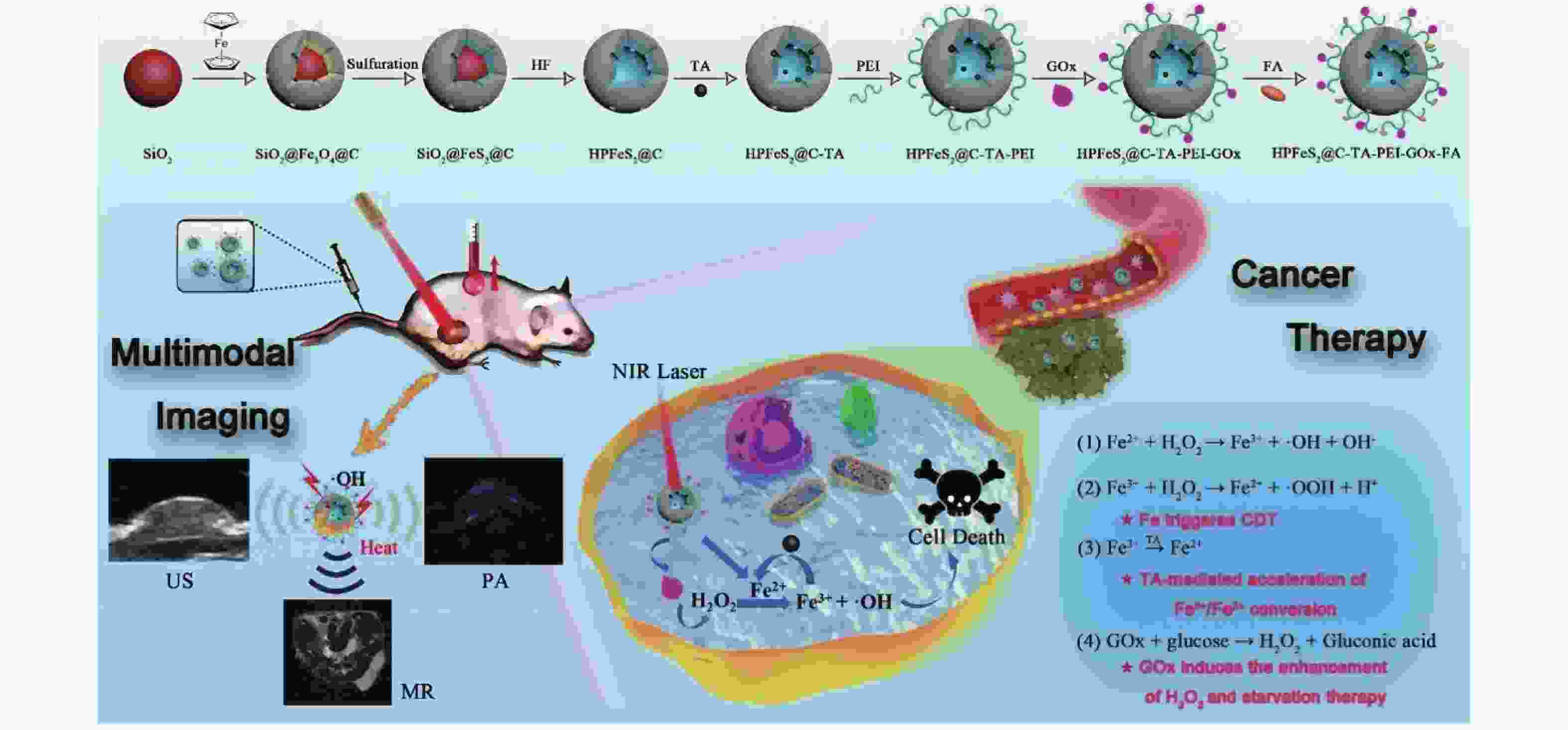
图 1 HPFeS2@C-TA-PEI-GOx-FA的制备工艺及治疗机理[11]
Figure 1. Preparation process and therapeutic mechanism of HPFeS2@C-TA-PEI-GOx-FA
-
[1] ZHONG X, WANG X, CHENG L, et al. GSH‐depleted PtCu3 nanocages for chemodynamic‐ enhanced sonodynamic cancer therapy[J] . Advanced Functional Materials,2019,30(4):1907954. [2] ZHANG M, LIU X, LUO Q, et al. Tumor environment responsive degradable CuS@mSiO2@MnO2/DOX for MRI guided synergistic chemo-photothermal therapy and chemodynamic therapy[J] . Chemical Engineering Journal,2020,389:124450. doi: 10.1016/j.cej.2020.124450 [3] CHEN Q, FENG L, LIU J, et al. Intelligent albumin-MnO2 nanoparticles as pH-/H2O2 -responsive dissociable nanocarriers to modulate tumor hypoxia for effective combination therapy[J] . Advanced Materials,2016,28(33):7129 − 36. doi: 10.1002/adma.201601902 [4] 胡进明, 刘世勇. 用于化学动力学疗法的高分子纳米载体研究进展[J] . 中国科学(化学),2020,50(3):366 − 376. [5] ZHANG C, BU W, NI D, et al. Synthesis of iron anometallic glasses and their application in cancer therapy by a localized Fenton reaction[J] . Angewandte Chemie. International Edition in English,2016,55(6):2101 − 2106. doi: 10.1002/anie.201510031 [6] KIM J, CHO H R, JEON H, et al. Continuous O2-evolving MnFe2O4 nanoparticle-anchored mesoporous silica nanoparticles for efficient photodynamic therapy in hypoxic cancer[J] . Journal of the American Chemical Society,2017,139(32):10992 − 10995. doi: 10.1021/jacs.7b05559 [7] LI X, LEE D, HUANG J D, et al. Phthalocyanine-assembled nanodots as photosensitizers for highly efficient type Ⅰ photoreactions in photodynamic therapy[J] . Angewandte Chemie. International Edition in English,2018,57(31):9885 − 9890. doi: 10.1002/anie.201806551 [8] LAN G, NI K, VERONEAU S S, et al. Titanium-based nanoscale metal-organic framework for type Ⅰ photodynamic therapy[J] . Journal of the American Chemical Society,2019,141(10):4204 − 4208. doi: 10.1021/jacs.8b13804 [9] CHEN H, TIAN J, HE W, et al. H2O2-activatable and O2-evolving nanoparticles for highly efficient and selective photodynamic therapy against hypoxic tumor cells[J] . Journal of the American Chemical Society,2015,137(4):1539 − 1547. doi: 10.1021/ja511420n [10] HUANG L, LI Z, ZHAO Y, et al. Ultralow-power near infrared lamp light operable targeted organic nanoparticle photodynamic therapy[J] . Journal of the American Chemical Society,2016,138(44):14586 − 14591. doi: 10.1021/jacs.6b05390 [11] WU F, ZHANG Q, ZHANG M, et al. Hollow porous carbon coated FeS2-based nanocatalysts for multimodal imaging-guided photothermal, starvation, and triple-enhanced chemodynamic therapy of cancer[J] . ACS Applied Materials and Interfaces,2020,12(9):10142 − 10155. doi: 10.1021/acsami.0c00170 [12] 杨博文, 陈雨, 施剑林. 纳米酶在肿瘤催化诊疗方面的应用[J] . 生物化学与生物物理进展,2018,45(2):237 − 255. [13] CHEN Z, YIN J J, ZHOU Y T, et al. Dual enzyme-like activities of iron oxide nanoparticles and their implication for diminishing cytotoxicity[J] . ACS Nano,2012,6(5):4001 − 4012. doi: 10.1021/nn300291r [14] CIOLOBOC D, KENNEDY C, BOICE E N, et al. Trojan horse for light-triggered bifurcated production of singlet oxygen and Fenton-reactive iron within cancer cells[J] . Biomacromolecules,2018,19(1):178 − 187. doi: 10.1021/acs.biomac.7b01433 [15] CHEN Q, ZHOU J, CHEN Z, et al. Tumor-specific expansion of oxidative stress by glutathione depletion and use of a Fenton nanoagent for enhanced chemodynamic therapy[J] . ACS Applied Materials and Interfaces,2019,11(34):30551 − 30565. doi: 10.1021/acsami.9b09323 [16] TANG Z, ZHANG H, LIU Y, et al. Antiferromagnetic pyrite as the tumor microenvironment-mediated nanoplatform for self-ernhanced tumor imaging and therapy[J] . Advanced Materials,2017,29(47):1701683. doi: 10.1002/adma.201701683 [17] HUO M, WANG L, CHEN Y, et al. Tumor-selective catalytic nanomedicine by nanocatalyst delivery[J] . Nature Communications,2017,8(1):1 − 12. doi: 10.1038/s41467-016-0009-6 [18] LIU Y, WU J, JIN Y, et al. Copper(I) phosphide nanocrystals for in Situ Self-Generation Magnetic resonance imaging-guided photothermal-enhanced chemodynamic synergetic therapy resisting Ddeep-seated tumor[J] . Advanced Functional Materials,2019,29(50):1904678. doi: 10.1002/adfm.201904678 [19] POYTON M F, SENDECKI A M, CONG X, et al. Cu2+ binds to hosphatidylethanolamine and increases oxidation in lipid membranes[J] . Journal of the American Chemical Society,2016,138(5):1584 − 1590. doi: 10.1021/jacs.5b11561 [20] TANG Z, LIU Y, HE M, et al. Chemodynamic therapy: Tumour microenvironment-mediated Fenton and Fenton-like reactions[J] . Angewandte Chemie. International Edition in English,2019,58(4):946 − 956. doi: 10.1002/anie.201805664 [21] LIN L S, HUANG T, SONG J, et al. Synthesis of copper peroxide nanodots for H2O2 self-supplying chemodynamic therapy[J] . Journal of the American Chemical Society,2019,141(25):9937 − 9945. doi: 10.1021/jacs.9b03457 [22] WANG X, ZHONG X, ZHA Z, et al. Biodegradable CoS2 nanoclusters for photothermal-enhanced chemodynamic therapy[J] . Applied Materials Today,2020,18:100464. doi: 10.1016/j.apmt.2019.100464 [23] LIN L S, SONG J, SONG L, et al. Simultaneous Fenton-like ion delivery and glutathione depletion by MnO2-based nanoagent to enhance chemodynamic therapy[J] . Angewandte Chemie. International Edition in English,2018,57(18):4902 − 4906. doi: 10.1002/anie.201712027 [24] WANG H, BREMNER D H, WU K, et al. Platelet membrane biomimetic bufalin-loaded hollow MnO2 nanoparticles for MRI-guided chemo-chemodynamic combined therapy of cancer[J] . Chemical Engineering Journal,2020,382:122848. doi: 10.1016/j.cej.2019.122848 [25] LIU Y, ZHEN W, JIN L, et al. All-in-one theranostic nanoagent with enhanced reactive oxygen species generation and modulating tumor microenvironment ability for effective tumor eradication[J] . ACS Nano,2018,12(5):4886 − 4893. doi: 10.1021/acsnano.8b01893 [26] WANG Z, LIU B, SUN Q, et al. Fusiform-like copper(II)-based metal-organic framework through relief hypoxia and GSH-depletion Co-enhanced starvation and chemodynamic synergetic cancer therapy[J] . ACS Applied Materials and Interfaces,2020,12(15):17254 − 17267. doi: 10.1021/acsami.0c01539 [27] WANG X, LV F, LI T, et al. Electrospun micropatterned nanocomposites incorporated with Cu2S nanoflowers for skin tumor therapy and wound healing[J] . ACS Nano,2017,11(11):11337 − 11349. doi: 10.1021/acsnano.7b05858 [28] SHI Y, ZHANG J, HUANG H, et al. Fe-doped polyoxometalate as acid-aggregated nanoplatform for NIR-II photothermal-enhanced chemodynamic therapy[J] . Advanced Healthcare Materials,2020,9(9):2000005. doi: 10.1002/adhm.202000005 [29] YANG C, YOUNIS M R, ZHANG J, et al. Programmable NIR-II photothermal-enhanced starvation-primed chemodynamic therapy using glucose oxidase-functionalized ancient pigment nanosheets[J] . Small,2020,16(25):2001518. doi: 10.1002/smll.202001518 [30] WANG Y, AN L, LIN J, et al. A hollow Cu9S8 theranostic nanoplatform based on a combination of increased active sites and photothermal performance in enhanced chemodynamic therapy[J] . Chemical Engineering Journal,2020,385:123925. doi: 10.1016/j.cej.2019.123925 [31] WANG S, WANG Z, YU G, et al. Tumor-specific drug release and reactive oxygen species generation for cancer chemo/chemodynamic combination therapy[J] . Advanced Science,2019,6(5):1801986. doi: 10.1002/advs.201801986 [32] HANG L, LI H, ZHANG T, et al. Au@Prussian blue hybrid nanomaterial synergy with a chemotherapeutic drug for tumor diagnosis and chemodynamic therapy[J] . ACS Applied Materials and Interfaces,2019,11(43):39493 − 39502. doi: 10.1021/acsami.9b13470 [33] KANKALA R K, TSAI P Y, KUTHATI Y, et al. Overcoming multidrug resistance through co-delivery of ROS-generating nano-machinery in cancer therapeutics[J] . Journal of Materials Chemistry B,2017,5(7):1507 − 1517. doi: 10.1039/C6TB03146C [34] REN Z, SUN S, SUN R, et al. A metal-polyphenol-coordinated nanomedicine for synergistic cascade cancer chemotherapy and chemodynamic therapy[J] . Advanced Materials,2020,32(6):1906024. doi: 10.1002/adma.201906024 [35] LIU C, WANG D, ZHANG S, et al. Biodegradable biomimic copper/manganese silicate nanospheres for chemodynamic/photodynamic synergistic therapy with simultaneous glutathione depletion and hypoxia relief[J] . ACS Nano,2019,13(4):4267 − 4277. doi: 10.1021/acsnano.8b09387 [36] WANG P, LIANG C, ZHU J, et al. Manganese-Based nanoplatform as metal ion-enhanced ROS generator for combined chemodynamic/photodynamic therapy[J] . ACS Applied Materials and Interfaces,2019,11(44):41140 − 41147. doi: 10.1021/acsami.9b16617 [37] DONG S, XU J, JIA T, et al. Upconversion-mediated ZnFe2O4 nanoplatform for NIR-enhanced chemodynamic and photodynamic therapy[J] . Chemical Science,2019,10(15):4259 − 4271. doi: 10.1039/C9SC00387H [38] NI K, LAN G, LIN W. Nanoscale metal-organic frameworks generate reactive oxygen species for cancer therapy[J] . ACS Cent Sci,2020,6(6):861 − 868. doi: 10.1021/acscentsci.0c00397 [39] NI K, LAN G, SONG Y, et al. Biomimetic nanoscale metal–organic framework harnesses hypoxia for effective cancer radiotherapy and immunotherapy[J] . Chemical Science,2020,11(29):7641 − 7653. doi: 10.1039/D0SC01949F -




 下载:
下载: